
Local governments suing the company have demanded roughly $2.4 billion in damages.Įndo ended sales of Opana ER in 2017 after the Food & Drug Administration found it was often abused by people who crushed, dissolved and injected the medication. Almost all our crime is driven by drugs," he told NPR. "It's overdose deaths, it's people losing their jobs, losing their dignity. It has really ripped at the fabric of our community," said Barry Staubus, district attorney in Sullivan County Tennessee. They claim Endo's opioid medication Opana ER was marketed aggressively in the state without proper safeguards, contributing to high rates of addiction and death. Stranch's legal team filed a series of complaints with the court about Endo's conduct in the case, which led to the judge's order.Ī controversial opioid, a devastating epidemicĪt issue is a lawsuit filed in 2017 by county prosecutors and other local government officials in eastern Tennessee. " lawyers crossed the line and worked with the company to subvert the court's orders and then made false statements to the court about it," he said. In a statement emailed to NPR the company said it will appeal the judgement.īut Gerard Stranch, one of the attorneys suing the drugmaker on behalf of local governments in Tennessee, said Endo and its lawyers "conspired to hide the truth" over a period of years as the fact-finding phase of the trial proceeded. Moody in a judgment issued on April 6.Įndo denies wrongdoing. "It appears to the court that Endo and its attorneys, after delaying trial, have resorted to trying to improperly corrupt the record," wrote Chancellor E.G. The judge presiding over the civil trial also concluded the drugmaker and its attorneys made at least a dozen false statements during the pretrial fact-finding process. All Rights Reserved.A state judge in Tennessee ruled the drug company Endo Pharmaceuticals is liable for harm caused by their opioid drug, Opana ER.Ī state court in Tennessee has punished Endo Pharmaceuticals for improperly withholding a vast trove of documents relating to the sale and marketing of its opioid medication Opana ER. 
Contact the publisher to obtain permission before redistributing.Ĭopyright – Unless otherwise stated all contents of this website are © 2017 Pro Pharma Communications International. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Available from: Permission granted to reproduce for personal and non-commercial use only. GaBI Online - Generics and Biosimilars Initiative. 
The company bought Par in 2015 for the significant sum of US$8 billion and recently had to lose 375 employees and stop work to develop a new pain medication. The more affordable generic form of Zetia may also take sales from more expensive anti-cholesterol drugs such as Praluent (alirocumab) and Repatha (evolocumab), produced by Sanofi and Amgen, respectively.Īlthough this may be concerning news for Merck, for Endo the Zetia generics will provide an opportunity for the company to boost its revenue following recent economic troubles. Combined, Zetia and Vytorin are expected to generate almost US$2 billion in 2017.Īnalysts from Barclays said loss of Zetia exclusivity will harm Merck’s earnings more than its sales, while Goldman Sachs commented that much of the recent growth in income from the drug has been due to price rises. The combination of ezetimibe with a statin has been shown to reduce cardiovascular risk in patients with acute coronary syndrome, where reduced blood flow through the coronary arteries affects the function of the heart. The launch could also threaten sales of Merck’s Vytorin, which is a combination of Zetia and the statin simvastatin (Zocor). Under US Food and Drug Administration (FDA) rules, Endo will have exclusive rights to the generic for the first six months, after which time Merck’s Zetia sales are likely to decline further as more generics makers enter the market. Working in partnership with Glenmark Pharmaceuticals, Endo began shipping 10 mg ezetimibe tablets on 12 December 2016. The drug is now threatened by its first generic version, produced by Par Pharmaceutical (Par), an operating company of Dublin’s Endo International (Endo). 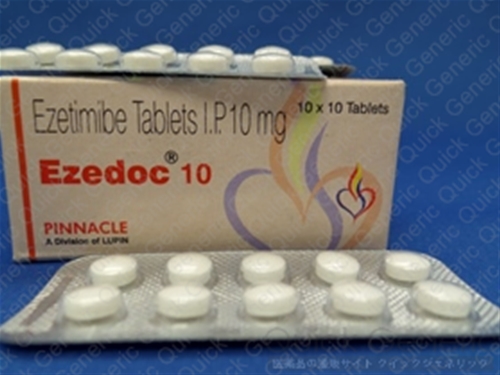
Zetia (ezetimibe), a blockbuster anti-cholesterol drug originally patented by Merck & Co (Merck) with US sales over US$2.6 billion for the year ending 30 September 2016, has come to the end of its patent protection.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
